Evidence of a new type of protein-protein interaction:
desensitized actomyosin blocks Ca2+-sensitivity of the natural one. A possible
model for an intracellular signalling system related to actin filaments
Vladimir V. Matveev
Laboratory of Cell Physiology, Institute of
Cytology, Russian Academy of Sciences,
Tikhoretsky Ave 4, St. Petersburg 194064, Russia and
Full text [PDF]
Abstract. Actin filaments are certainly believed to function as
an intracellular signalling system; however, this is not confirmed by direct
evidence. We used a two-layer actomyosin gel with a concen tration gradient of
the troponin-tropomyosin complex (TT-complex, Ca2+-sensitive system)
between the two layers. To prepare one layer of the system, natural actomyosin (nAM)
rich in TT-complex was used. To prepare the second layer, we used desensitized
actomyosin (dAM) without the complex. All experimental studies were
made in medium with a low ionic strength. Two phenomena were ob served: (1)
dAM blocks Ca2+-sensitivity of nAM when the dAM
weight portion in the system (as well as in mixed nAM+dAM
suspension) reaches 40% and more; further increase of the dAM portion
does not affect the Ca2+-sensitivity; (2) it was electrophoretically
shown that a rapid diffusion of the TT-complex from nAM gel into the
dAM gel took place. The apparent diffusion coefficient for the
TT-complex in dAM gel is about (1-4)Ę10-4 cm2/sec,
i.e. three orders higher than the same values for protein diffusion in water.
The cytoskeleton and, particularly, actin filaments are
believed to function as an intracellular signalling system (by signal we mean
any cellular event which has a regulatory significance). Edelman (1) was the
first to suggest participation of actin filaments in the transfer of signals
from the plasma membrane to the cytoplasm and nucleus.
To elucidate the mechanism of transfer of a signal related to elements of the
cytoskeleton, experiments with protein model systems reproducing some important
features of the cytoplasm can be useful. Such a convenient model system can be
actomyosin. Natural actomyosin (nAM) contains a regulatory
troponin-tropomyosin complex (TT-complex), whose content may be easily changed
by washing out actomyosin with a solution of a low ionic strength and alkaline
pH. After removal of the TT-complex, the actomyosin superprecipitation by
addition of MgATP becomes Ca2+-independent, i.e., actomyosin becomes
desensi tized (dAM). Addition of the TT-complex to dAM results
in recovery of Ca2+-sensitivity (2). Therefore, after removal of the
TT-complex from actomyosin, its binding sites still are able to bind again the
regulatory complex. Thus, the use of actomyosin preparations with different
contents of TT-complex can produce gels with a concentration gradient of the
actin-binding proteins.
It should be emphasized that the TT-complex is strongly bound to actin filaments
at physiological ionic strength and pH and serves as a Ca2+ trigger
of muscle contraction (3,4). Hence, the function and distribution of this
regulatory complex can be evaluated not only by analysis of protein content in
various areas of the gel, but also by measurement of its functional parameter,
Ca2+-sensitivity of the reaction of actomyosin superprecipitation.
The goal of the present work was to study the interaction of actomyosin gels
with different contents of TT-complex to detect possible redistribution of the
TT-complex as a result of its diffusion, translocation, transport, etc.
Materials and Methods
Isolation of natural actomyosin (nAM)
from rabbit skeletal muscles was performed at 4-6░C, as described by Haga et al.
(5), with minor modifications. The time of the extraction was 15-20 h. The
actomyosin gel (about 30 mg/ml of protein) with 50 mM KC1 was diluted by the
same volume of cold glycerol and stored at -20░C (6). Prior to the experiments,
the glycerinated actomyosin was washed free of glycerol with a solution
containing 90 mM KCl and 14 mM Na-K-phosphate buffer, pH 6.7. After two washings
by tenfold volumes of this phosphate buffer (PB), nAM was dissolved in
0.5 M KC1 and centrifuged for 20 min at 10,000 g. The supernatant was diluted
with 9 volumes of cold deionized water, and the collected precipitate was washed
twice with PB and then resuspended in PB.
Preparation of desensitized actomyosin (dAM)
was done by washing nAM at 4-6░C in pure water containing a minimal
amount of Tris-buffer, pH 8.5-9.0 (2). 15-20 ml aliquots of the stored
glycerolized actomyosin preparation were placed into every 100-ml centrifuge
test tube, and water was added up to the volume of 70 ml. After the second or
third washing, with centrifugation for 20 min at 10,000 g, the actomyosin gel
swelled (as a result of desalination) to such a degree that it could no longer
be precipitated. After this step, prior to every centrifugation, 1-2 drops of 3
M KCl were added to every tube so that the actomyosin gel volume was about 50%
of the suspension volume after centrifugation. After each centrifugation, the
supernatant was replaced by fresh pure water. This procedure was repeated 10
times. The above procedure was confirmed electrophoretically to result in the
nearly complete removal of the TT-complex and in the complete elimination of the
Ca2+-sensitivity. The sensitivity was effectively restored by
addition of the TT-complex isolated as described by Spudich and Watt (7) to such
dAM preparations. After washing, dAM was centrifuged in 0.5 M
KCl and precipitated by dilution with 10 volumes of water. The precipitate was
washed twice with PB and then resuspended in the third portion of the buffer.
Superprecipitation of nAM, dAM, and their
mixtures was carried out at 20░C in PB con taining 0.5 mM Ca2+ or 0.5 mM EGTA
(ethylene glycol-bis(ß-aminoethyl ether) N,N,N',N'-tetraacetic acid) and 0.25 mM
MgATP at a protein concentration of 0.1 mg/ml. The reaction of
superprecipitation was recorded by the method of 90░-angle light scattering at
450 nm, using a Specol spectrometer (Carl Zeiss, Jena, Germany) with a built-in
magnetic mixer for continuous stirring of the samples in the course of
superprecipitation. For the superprecipitation, 30-ml cells were used; the
volume of samples was 20 ml.
After addition of MgATP to the actomyosin suspension, the light scattering began
to fall, reaching the minimum steady-state level after 1-2 min (for nAM)
or after 5-7 min (for dAM). For nAM preparations, the
decreasing value of the light scattering varied depending on the presence of Ca2+
or EGTA in the solution. This sensitivity to Ca2+ (Sca)
was ex pressed quantitatively as a light scattering decrease (LSD): LSD =10-1,
in the presence both of 0.5 mM Ca2+ (LSDca) and of 0.5 mM
EGTA (LSDegta), where 10 is the initial and 1, the final level of the
light scattering (in arbitrary units). So Sca = LSDca/LSDegta.
As a rule, three measurements of Sca were done for each actomyosin
sample.
The Sca values for dAM preparations were close to 1, which
indicates that the superprecipitation of these preparations did not depend on Ca2+
and, accordingly, that no significant amount of the regulatory TT-complex
remained in dAM. The Sca values for nAM prepara
tions were more than 1. This method of measurement of Ca2+-sensitivity
is described in de tail in our earlier work (6).
Electrophoresis in polyacrylamide gels in the presence of
sodium dodecyi sulphate (disc-SDS-electrophoresis) was performed by the method
of Sheludko (8), with some modifica tions for block-electrophoresis. The gel
components were from Reanal (Hungary). Their composition in the separation gel,
unless otherwise specified, was the following: 0.43 M Tris-HCl (tris(oxymethyl)aminomethane),
pH 8.9, 12% acrylamide (AA), and 0.3% N,N'-methylene-bis-acrylamide (MBA), in
the concentration gel: 0.05 M Tris-HCl, pH 6.8, 2.5% AA, 0.6% MBA, 20% sucrose,
and 2% sodium dodecyl sulphate. The electrode solutions contained 0.6 M glycine
and 0.033 M Tris-buffer, pH 8.3; sodium dodecyl sulphate was added to the upper
electrode solution up to a concentration of 0.1%. Actomyosin samples were
solubilized for 5 min in a boiling water bath in a solution containing 50%
glycerol, 5% sodium dodecyl sulphate, 50 mM dithiothreitol (Serva, Germany), and
25 mM EDTA (ethyienediaminetetraacetic acid), pH 7.0. The gels were fixed by a
mixture of ethanol-CH3COOH-H2O (1:3:6) and then stained
with 0.125% Coomassie brilliant blue G-250 (Serva, Germany) dissolved in the
fixation mixture. After the dye excess had been removed from the gels with 7%
acetic acid, densities of protein bands were measured three times, us ing an
Elscript-3 densitometer (Hirschmann, Germany). The relative contents of
TT-complex proteins were expressed as percentage to actin.
Protein concentration was determined by the micro-biuret method
(9), using bovine se rum albumin as a standard.
Formation of two-layer gels. To form an actomyosin gel with
non-uniform distribution of the TT-complex, a two-layer system was prepared in
10-ml centrifuge tubes (Figure 1), using the following steps. First, 30 mg of
the dAM in suspension were placed in each tube and precipitated by
centrifugation for 20 min at 10,000 g (with the final protein concentration of
30-40 mg/ml in the precipitate). Then the supernatant was removed; different
amounts of the nAM suspension were added very carefully, and the
centrifugation was repeated. The second centrifugation produced a two-layer
system (Figure 1) with the overlying supernatant that remained in the tubes
during the entire experiment. In order to test deformability of the interlayer
surface during the second layer formation, the surface of the first layer was
marked with the dye neutral red or with colloidal carbon. No deformation was
detected after the second centrifugation. So the possibility of a mixture of
different actomyosin preparations can be ruled out.
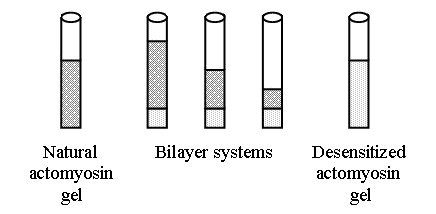
FIGURE 1. A diagrammatic sketch of the two-layer actomyosin systems used, each with a different ratio of natural (nAM) and desensitized (dAM) actomyosin. The sizes of the layers are overesti mated to better demonstrate the arrangement of the experiments.
In separate experiments, it was found that the nAM layer (in the
two-layer system with the nAM/dAM ratio 60:40, respectively)
lost its Ca2+-sensitivity as early as after 1-3 min. But to insure
the steady-state conditions, we used an overnight incubation both for the
two-layer systems and for two-component (n- and dAM)
suspensions. However, the apparent diffusion coefficient was determined during
much shorter time periods.
After formation of the two-layer systems, the tubes were chilled for 15-20 h at
4-6░C be fore the electrophoresis and superprecipitation. After incubation,
minimal, about 2 mg, samples of actomyosin were carefully taken exactly from the
surface of the upper layer of the two-layer system for measurements of the Ca2+-sensitivity
and electrophoretic analysis. The stock nAM and dAM
preparations used to form the two-layer system served as control.
To determine the apparent diffusion coefficient (ADC) for the
TT-complex (tropomyosin was used as a marker of the complex), the nAM-dAM
interaction was performed in thin polyvinyl chloride tubes that were chosen so
that the outside diameter of one tube corre sponded to the inside diameter of
the other. This made it possible to form a very tight, mechanically strong
connection. Tubes with the internal diameters of 3 and 6 mm were used; they
formed 2-3-cm-long gels; one end of each tube was sealed.
The thin tubes were filled with nAM, the thick ones, with dAM.
The filling of the tubes with actomyosin gel was carried out by the same
centrifugation procedure, as in the preparation of the two-layer systems (10,000
g, 20 min). After formation of the precipitate, the tubes were cut at the level
of the precipitate/supernatant line and connected to each other very carefully.
The protein contamination from the surface of the joined tubes was thoroughly
removed.
Several joined tubes were then incubated at 4-6░C. These two-tube systems were
quickly frozen on ice at -20░C after different incubation time (from 3 min to 1
h). Then the thin and thick tubes were separated into two sections, left and
right, respectively, 1 mm from the point of gel contact. The segment isolated
after these first two cuts was discarded.
The separated tubes were cut into 5 mm slices (resulting in 4-6 segments). After
each cut, the cutting tool was cleaned of protein contamination. Protein from
each segment was solubilized and subjected to electrophoresis. This allowed for
the detection of the time-dependent tropomyosin decrease in the nAM gel and the
tropomyosin increase in dAM. The TT-complex diffusion was studied at a
nAM/dAM weight ratio 1:4. The ADC was calculated as described
by Chang (10).
Statistics. Results of the experiments were expressed as means
▒ SEM. The differences were considered statistically significant at P < 0.05,
using Student's t-test.
Results
Figure 2 (curve A) illustrates typical results of the experiments with the two-layer systems. The control nAM preparation definitely demonstrated a high sensitivity to Ca2+ (curve A, the left end, 0% of dAM), while the other control preparation, dAM, was completely desensitized (curve A, the right end, 100% of dAM). In the two-layer system, an increase in the proportion of the dAM weight within narrow limits from 20 to 40% (20% range) resulted in the complete loss of Ca2+-sensitivity in nAM (the top layer).
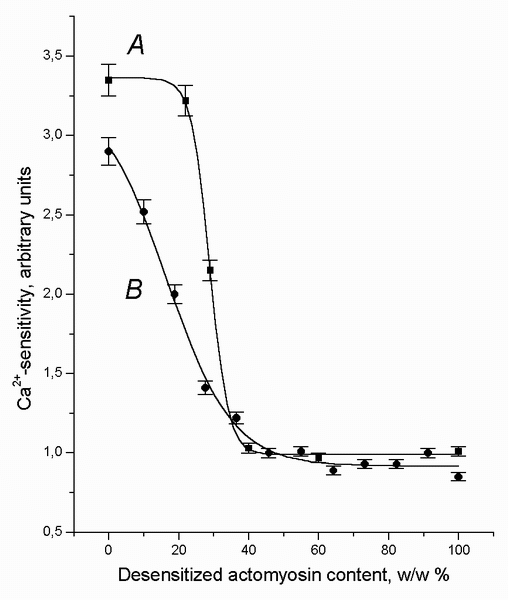
FIGURE 2. The Ca2+-sensitivity of the natural actomyosin (nAM), A, arranged as the upper part of the two-layer system (See Figure 1), and the Ca2+-sensitivity of the two-component suspension mixtures (1 mg of protein per ml) of nAM with desensitized actomyosin (dAM.), B, following the 15-20 h incubation (at 4-6░C) plotted as a function of the weight portion of the dAM. The value "0" on the X-axis means nAM preparation alone. The value "100" on the X-axis means dAM preparation alone. The value "1" on the Y-axis means the absence of the Ca2+-sensitivity. The data points are the means ▒ SEM from triplicate assays.
As seen from the shape of the curve A, the degree of the TT-complex
redistribution depends on the weight ratio of the layers, therefore, diffusion
of the minor components is triggered by the weight ratio, rather than by the
existence of the gradient alone. This two-layer system as a whole initiates the
protein diffusion at a definite nAM/dAM threshold (critical)
ratio.
This "threshold" hypothesis is confirmed by the data shown in Figure 3. As seen
from Figure 2, the curve describing the dependence of Ca2+-sensitivity
on the weight portion of dAM (without the points for 0 and 100% dAM
because these points do not reflect interactions in two-layer systems) looks
like a hyperbola. After transformation of the data of Figure 2 into the
coordinates of Figure 3, this dependence becomes linear. The linear character of
the relationships demonstrated by Figure 3 is statistically significant for the
two-layer system at the 95% confidence level (correlation coefficient = 0.989, P
< 0.05) and for the two-component suspension, at the 99% confidence level
(correlation coefficient = 0.994, P < 0.01). This linearity proves the
hyperbolic character of the relationships seen in Figure 2 for interacting
actomyosin preparations. Therefore, we can regard the X-axis asymptote (see the
legend for Figure 3) for each curve as a corresponding nAM/dAM
threshold ratio. The threshold value for dAM content to initiate
blockage of Ca2+-sensitivity of nAM was calculated to be 18
▒ 5% for the two-layer system and 9 ▒ 2%, for the two-component suspension.
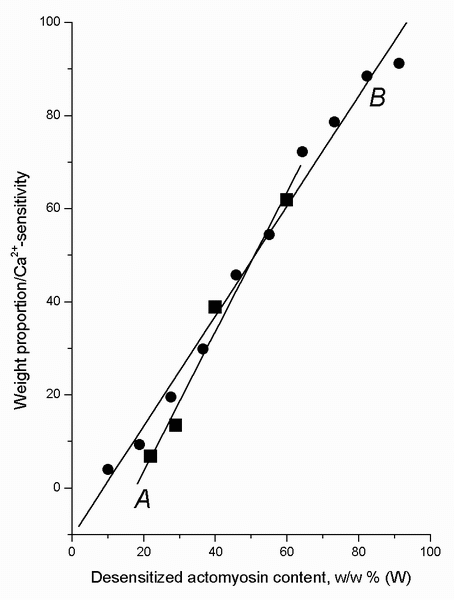
FIGURE 3. The linear representation of the data from Figure 2 for natural (nAM) + desensitized actomyosin (dAM) preparations in a form of two-layer systems, A, and as two-component suspension, B. The experimental results (except for the data for individual nAM and dAM preparations, in which the studied interaction does not occur) are well fitted by the linear model: W/S = a + bĘW, where W is the weight portion (in %) of dAM; S is the Ca2+-sensitivity (in arbitrary units) of nAM (as a part of the two-layer system) or of the nAM + dAM suspension mixtures. The parameters of the equation for the suspensions are: W/S = -10.4(▒2.7) + 1.18(▒0.05) ĘW, correlation coefficient = 0.994 (P-value < 0.01); for the two-layer systems: W/S = -26.4(▒6.6) + 1.50(▒0.16) ĘW, correlation coefficient = 0.989 (P-value < 0.05). The statistically significant linearity of the relationships indicates that the data both for the two-layer systems and for the nAM + dAM suspension mixtures demonstrate a hyperbolic relation between S and W values. If so, it is possible to calculate the X-axis asymptote for each curve in Figure 2 as a value for the threshold nAM/dAM ratio in accordance with the relation: Threshold = a/b, where a and b are parameters of the linear model mentioned above (the absolute value of a is calculated). The following values for the thresholds were obtained: 9▒2% for the suspensions, and 18▒5% for the two-layer systems (means ▒ SEM). These thresholds are the levels of dAM (in %, w/w) when the redistribution of TT-complex begins in the systems.
As seen from the shape of the curve A, the degree of the TT-complex
redistribution depends on the weight ratio of the layers, therefore, diffusion
of the minor components is triggered by the weight ratio, rather than by the
existence of the gradient alone. This two-layer system as a whole initiates the
protein diffusion at a definite nAM/dAM threshold (critical)
ratio.
This "threshold" hypothesis is confirmed by the data shown in Figure 3. As seen
from Figure 2, the curve describing the dependence of Ca2+-sensitivity
on the weight portion of dAM (without the points for 0 and 100% dAM
because these points do not reflect interactions in two-layer systems) looks
like a hyperbola. After transformation of the data of Figure 2 into the
coordinates of Figure 3, this dependence becomes linear. The linear char acter
of the relationships demonstrated by Figure 3 is statistically significant for
the two-layer system at the 95% confidence level (correlation coefficient =
0.989, P < 0.05) and for the two-component suspension, at the 99% confidence
level (correlation coefficient = 0.994, P < 0.01). This linearity proves the
hyperbolic character of the relationships seen in Figure 2 for interacting
actomyosin preparations. Therefore, we can regard the X-axis asymptote (see the
legend for Figure 3) for each curve as a corresponding nAM/dAM
thresh old ratio. The threshold value for dAM content to initiate
blockage of Ca2+-sensitivity of nAM was calculated to be 18
▒ 5% for the two-layer system and 9 ▒ 2%, for the two-component suspension.
Since nAM (the top layer) lost its Ca2+-sensitivity, the
question arises as to whether the change in the content of the TT-complex
actually takes place in this layer of the two-layer system under the conditions
of our experiment. To answer this question, several two-layer systems with
different nAM/dAM ratios were formed (Figure 1), and the
content of the regulatory proteins in the top layer of the systems was
determined (after 10-15 h of incubation) by the method of electrophoresis.
Figure 4 shows that the content of all the studied components of the regulatory
complex fall sharply when the weight proportion of dAM exceeded a
certain threshold.
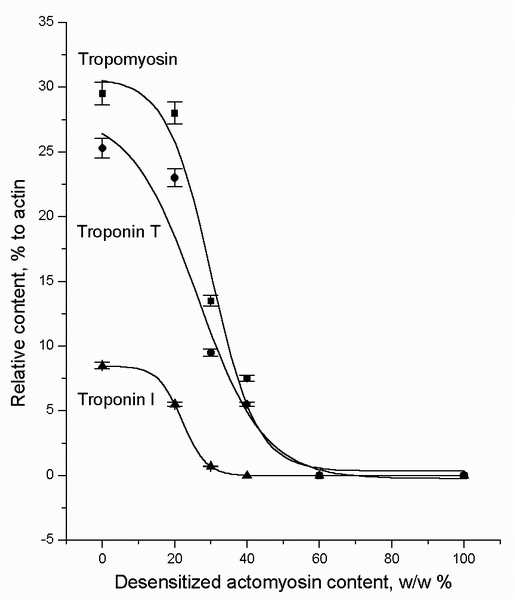
FIGURE 4. The tropomyosin, troponin T, and troponin I contents in the natural actomyosin (nAM) arranged as the top layer of the two-layer systems (see Figure 1), plotted as a function of the weight portion of the desensitized actomyosin (dAM) in the lower layer after 15-20 h of incubation (at 4-6░C). These data indicate that the TT-complex diffuses from the nAM to the dAM layer in the two-layer systems. The value "0" on the X-axis means nAM preparation alone. The value "100" on the X-axis means dAM preparation alone. The data points are the means ▒ SEM from triplicate assays.
Figure 2 into the coordinates of Figure 3, this dependence becomes linear. The
linear character of the relationships demonstrated by Figure 3 is statistically
significant for the two-layer system at the 95% confidence level (correlation
coefficient = 0.989, P < 0.05) and for the two-component suspension, at the 99%
confidence level (correlation coefficient = 0.994, P < 0.01). This linearity
proves the hyperbolic character of the relationships seen in Figure 2 for
interacting actomyosin preparations. Therefore, we can regard the X-axis
asymptote (see the legend for Figure 3) for each curve as a corresponding nAM/dAM
thresh old ratio. The threshold value for dAM content to initiate
blockage of Ca2+-sensitivity of nAM was calculated to be 18
▒ 5% for the two-layer system and 9 ▒ 2%, for the two-component suspension.
Since nAM (the top layer) lost its Ca2+-sensitivity, the question arises as to
whether the change in the content of the TT-complex actually takes place in this
layer of the two-layer system under the conditions of our experiment. To answer
this question, several two-layer systems with different nAM/dAM
ratios were formed (Figure 1), and the content of the regulatory proteins in the
top layer of the systems was determined (after 10-15 h of incubation) by the
method of electrophoresis. Figure 4 shows that the content of all the studied
components of the regulatory complex fall sharply when the weight proportion of
dAM exceeded a certain threshold.
With respect to the Ca2+-sensitivity changes, similar results were
also obtained for mixed nAM and dAM suspensions containing
different proportions of dAM (Figure 2, curve B) (e.g., with a final
protein concentration of 1 mg/ml, each ml in the 40% dAM mixture would
contain 0.4 mg of dAM and 0.6 mg of nAM). Like in the case
when the dAM weight proportion in the two-layer systems reached the 40%
level, the two-component mixture completely lost Ca2+-sensitivity at
the same level of dAM, although it consisted of 60% of nAM
containing a significant amount of the native TT-complex. The cooperative
character of the B curve (Figure 2) might indicate an interaction
between the two types of actomyosin preparations, otherwise the relationship
between the Ca2+-sensitivity and dAM content would have had
a linear character.
As the TT-complex is the receptor of Ca2+-, the Ca2+-dependence
of the nAM-dAM interaction was studied. These sets of
experiments were performed as were other experiments with two-component
suspensions but with the following differences: incubation time was 1 h;
incubation medium contained 0.5 mM EGTA or 0.5 mM Ca2+. When Ca2+-sensitivity
of the preparations with EGTA was measured, excess Ca2+ was injected
about 1-2 sec prior to the MgATP injection. Data plotted in Figure 5 show that
free Ca2+ enhances the inactivation of the Ca2+-sensitive
system (by dAM) but does not affect the regression slope of the Ca2+-sensitivity
versus dAM: the close-to-linear parts of the curves are almost
parallel.
Our measurements showed that the tropomyosin (in fact, the TT-complex) ADC
(apparent diffusion coefficient) varied within the limits of 4.2-4.4Ę10-4 cm2/sec
in the presence of Ca2+ (0.5 mM) and within the limits of
2.7-4.2Ę10-4 cm2/sec in the presence of EDTA (0.5 mM). Thus, it is
evident that divalent cations (Ca2+ and Mg2+) do not
significantly affect the rate of the tropomyosin/TT-complex diffusion under the
conditions of our experiments.
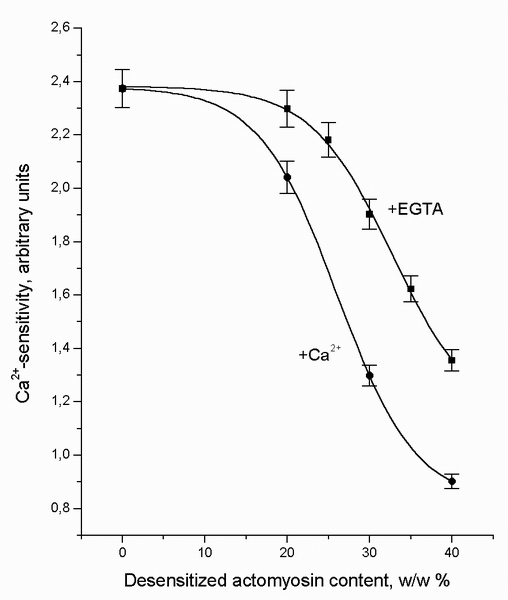
FIGURE 5. The Ca2+-sensitivity of the two-component suspension mixtures (1 mg of protein per ml) of natural actomyosin (nAM) with desensitized actomyosin (dAM) plotted as a function of the weight portion of the dAM in the mixture after one-hour incubation (at 4-6░C) in the presence of 0.5 mM EGTA or 0.5 mM Ca2+. When Ca2+-sensitivity of the preparations with EGTA was measured, excess of Ca2+ was injected about 1-2 sec prior to the MgATP injection. The value "0" on the X-axis means 100% nAM content in the suspension. The data points are the means ▒ SEM from triplicate assays.
Discussion
An understanding of the nature of the Ca2+-sensitivity of actomyosin
superprecipitation is essential to interpreting the results obtained in this
work. The TT-complex has been shown to be the final component of the
excitation-contraction coupling system (11). This complex also determines
Ca2+-sensitivity of the natural actomyosin superprecipitation (2-4). Its removal
results in the loss of Ca2+-sensitivity in actomyosin, but addition
of the TT-complex to a reconstituted actomyosin that contains no other proteins
but actin and myosin produces Ca2+-sensitivity of its
superprecipitation.
In this connection, of interest are our data using the two-layer gel (Figure 1).
We found a new type of interaction of the contractile proteins both in two-layer
systems and in suspen sion mixtures. The nAM preparations studied had a
high Ca2+-sensitivity (3.3 arbitrary units) before interaction with
the dAM layer (Figure 2). However, after the nAM gel con tacts with the dAM
gel, the Ca2+-sensitivity of nAM (the top layer) begins to
depend on the dAM/nAM weight ratio in the two-layer system;
this dependence has a threshold and cooperative pattern: nAM completely
loses Ca2+-sensitivity within the narrow limits of this ratio
changes, from 22/78 to 40/60. This suggests that nAM, the top layer of
the two-layer gel, loses the regulatory TT-complex.
The amount of the Ca2+-sensitive complex in the top layer is actually
reduced with an in crease in the amount of dAM in the system (Figure
4). At the dAM/nAM ratio = 4/6, the tropomyosin amount, for
example, in the top layer decreases more than 3 times. The amount of other
regulatory proteins also falls markedly. The above-mentioned ratio, 4/6, is the
limit, after which the actomyosin preparations lose completely their Ca2+-sensitivity.
The change in the Ca2+-sensitivity of nAM in the two-layer
system has a definite cooperative character that indicates that two actomyosin
preparations somehow interact with each other and this interaction spreads far
from the line of contact of the two gels.
It is clear that the nAM-dAM interaction begins in a zone of
contact of the two gels, but then this process involves the entire system. Thus,
the molecular events in the protein sys tem studied appear spatially
interconnected.
The mechanism of such changes requires the ability of the minor components of
actomyosin gel to move inside the gel. This conclusion is based on the data
presented in Figure 4. These data indicate that in the bulk of the two-layer
gel, after the start of contact was made, the spatial redistribution (diffusion)
of the TT-complex molecules begins to be visible as evidenced by the minor
components leaving the top layer of the system (nAM) to the point where
nAM loses its Ca2+-sensitivity. It is highly probable that
the structural basis for the diffusion of the TT-complex is F-actin, because
tropomyosin and the complex strongly bind to it (4), and the sigmoid character
of the interaction is caused by the cooperative properties of actin filaments
and proteins associated with them (12,13).
The nAM-dAM interaction also occurs in the two-component
suspensions (Figure 2, curve B). It is seen that with a decrease in the Ca2+-sensitive
nAM fraction, the decrease in the Ca2+-sensitivity of the
whole mixture is not linear; this relationship might indicate a more complex,
unexpected interaction. In fact, as soon as the nAM content falls below
60%, the Ca2+-sensitivity of the mixture disappears completely.
The interaction of nAM with dAM in the presence of free Ca2+
causes a greater decrease in the Ca2+-sensitivity (Figure 5). This is
definitely reasonable, considering multiple Ca2+ effects on the
properties and structure of contractile and regulatory proteins and on their
complexes (4).
It is well known that Ca2+, in addition to its effect on the
structure and properties of the TT-complex (14), enhances the cooperativity and
changes some other properties of actin filaments (13,15) as well as reduces by
twice or more the actin affinity to the TT-complex (16). The latter effect is of
interest, as the weakening of the F-actin-TT-complex interaction may facilitate
diffusion of the complex. In the presence of EGTA, actin filaments are more
rigid than in the presence of Ca2+ (17); but in the presence of Ca2+,
the regulated actin (F-actin + TT-complex) and F-actin alone become very
flexible (18,19). From our point of view, the flexibility of actin filaments is
one of the factors modulating the rate of the de crease or loss of the Ca2+-sensitivity
after the beginning of the nAM-dAM interaction: the flexible
filament facilitates the diffusion process and thus accelerates redistribution
of the TT-complex.
It is interesting to compare the TT-complex apparent diffusion coefficient (ADC)
obtained in this work for the condensed actomyosin gel (at 4-6░C) with that for
diffusion of other substances in water (at 25░C) (10): 1.10Ę10-5,
ethanol; 1.18Ę10-5, urea; 4.6Ę10-6, sucrose; 6.9Ę10-7,
hemoglobin; 13.3Ęl0-7 cm2/sec, myoglobin. It should be
noted again that in our experiments, tropomyosin was used as a protein marker
for the TT-complex. It might well be that the ADC obtained for "tropomyosin" is
in fact the coefficient for TT-complex.
Thus, the TT-complex ADC measured at a lower temperature and in much more
viscous, almost solid, medium than water has an ADC value that is one order of
magnitude higher than that for small molecules of ethanol and urea and three
orders of magnitude higher than that of hemoglobin and myoglobin. The diffusion
coefficients for other proteins (lysozyme, ovalbumin, bovine serum albumin), and
tobacco virus, as well as for DNA, also appeared to be three orders of magnitude
lower (20) than that for TT-complex. Hence, if the TT-complex diffused in the
water inside the actomyosin gel, its diffusion coefficient would be much lower.
In other words, the mechanism of the fast diffusion does not allow TT-complex to
dissociate from actomyosin.
On the other hand, protein diffusion in the cytoplasm (condensed protein system)
is much slower than in aqueous solutions. Thus, the ADCs for proteins in
cytoplasm are: 1.0Ę10-8 cm2/sec, bovine serum albumin (human fibroblasts, at
22░C) (21); 5.7Ę10-8 cm2/sec, aldolase (Swiss 3T3 cells, at 36░C)
(22); 2-3Ę10-9 cm2/sec, G-actin (embryonic chicken gizzard
cells, at 36░C) (23); 1.2Ę10-7 cm2/sec, myoglobin (muscle
fibers, at 22░C) (24). As a rule, the protein mobility in the cytoplasm is much
slower than in aqueous solutions. This indicates that the mobility inside the
cell is limited by interaction of diffusible proteins with the cytoplasmic
matrix (25).
It is interesting to discuss which properties of F-actin can promote the
"sliding" diffusion of regulatory proteins bound to it: (1) tropomyosin bound to
actin has an ability for lateral movement (26-30); (2) F-actin itself can
facilitate diffusion of the TT-complex by sponta neous bends (18,19), a "twist"
of monomeric units in coordination with each other (31), and rotational
movements of actin globules (32).
Our findings seem to indicate that the actomyosin gel can be a convenient model
for studies on processes of intracellular signalling systems related to actin
filaments. This model (a protein gel based on F-actin) is most likely to provide
promising opportunities for studies of conditions required for protein diffusion
in condensed protein systems like the cytoplasm. The phenomenon of the fast
diffusion of TT-complex recalls findings of a higher mobility of cations bound
to polyanions in comparison with the their lower motility in water (for review
see 33). These data indicate that the association does not necessarily lead to a
pronounced reduction in mobility.
So the data obtained can be summarized in the following words. Natural
actomyosin enriched in the troponin-tropomyosin complex interacts specifically
(by a mechanism still un known) with desensitized actomyosin free of the
regulatory complex. This results in (i) loss of Ca2+-sensitivity in
natural actomyosin placed both as a layer of the two-layer system and as a
component of its mixture with desensitized actomyosin, and (ii) redistribution
of the TT-complex in the actomyosin gel. The unknown mechanisms of these effects
may play an important role in the living cell.
The author wishes to thank sincerely Dr. A.A. Vereninov for helpful support of
this study. The author is also grateful to Dr. L.Z. Pevzner for criticism and to
Mr. J. Picheny for his assistance.
REFERENCES
1. Edelman, G.M. (1976) J. Biochem. (Tokyo) 79:1P.
2. Schaub, M.C. and Perry, S.V. (1969) Biochem. J. 115:993.
3. Farah, C.S. and Reinach, F.C. (1995) FASEB J. 9:755.
4. Marston, S. (1995) Int. J. Biochem. Cell Biol. 27:97.
5. Haga, T., Maruyama, K. and Noda, H. (1965) Biochim. Biophys. Acta 94:226.
6. Matveev, V.V. and Suzdalskaya, I.P. (1980) Tsitologiia 22:676.
7. Spudich, J.A. and Watt, S. (1971) J. Biol. Chem. 246:4866.
8. Sheludko, N.S.(1975) Tsitologiia 17:1148.
9. Itzaki, R.F. and Gill, D.M. (1964) Analyt. Biochem. 9:401.
10. Chang, R. (1977) Physical Chemistry with Application to Biological Systems.
Macmillan Publishing Co. Inc., New York.
11. Katz, A.M. (1995) Cardioscience 6:1.
12. Brandt, P.W., Diamond, M.S., Gluck, B., Kawai, M. and Schachat, F. (1984)
Carlsberg Res. Commun. 49:155.
13. Praser, I.D. and Marston, S.B. (1995) J. Biol. Chem. 270:7836.
14. Leavis, P.C. and Gergely, J. (1984) CRC Crit. Rev. Biochem. 16:235.
15. Popp.D. and Maeda.Y. (1993) J. Mol. Biol. 229:279.
16. Wegner, A. and Walsh, T.P. (1981) Biochemistry 20:5633.
17. Schwienbacher, C., Magri, E., Trombetta, G. and Grazi, E. (1995)
Biochemistry 34:1090.
18. Fujime, S. and Ishiwata, S. (1972) J. Mol. Biol. 62:251.
19. Ishiwata, S. and Fujime, S. (1972) J. Mol. Biol. 68:511.
20. Dubin, S.B., Lunacek, J.H. and Benedek, G.B. (1967) Proc. Natl. Acad. Sci.
USA. 57:1164.
21. Wojcieszyn, J.W., Schlegel, R.A., Wu, E.S. and Jacobson, K.A. (1981) Proc.
Natl. Acad. Sci. USA. 78:4407.
22. Pagliaro, L. and Taylor, D.L. (1988) J. Cell Biol. 107:981.
23. Kreis, T.E., Geiger, B. and Schlessinger, J. (1982) Cell 29:835.
24. Jurgens, K.D., Peters, T. and Gros, G. (1994) Proc. Natl. Acad. Sci. USA.
91:3829.
25. Gershon, N.D., Porter, K.R. and Trus, B.L. (1985) Proc. Natl. Acad. Sci.
USA. 82:5030.
26. Hanson, J., Lednev, V., O'Brien, E.J. and Bennet, P.M. (1972) Cold Sring
Harbor Symp. Quant. Biol. 37:311.
27. Haselgrove, J.C. (1972) Cold Sring Harbor Symp. Quant. Biol. 37:341.
28. Huxley, H.E. (1972) Cold Sring Harbor Symp. Quant. Biol. 37:361.
29. Parry, D.A.D. and Squire, J.M. (1973) J. Mol. Biol. 75:33.
30. Lehman, W., Craig, R. and Vibert, P. (1994) Nature 368:65.
31. Stokes, D.L. and DeRosier, D.J. (1987) J. Cell Biol. 104:1005.
32. Yanagida, T. and Oosawa, F. (1980) J. Mol. Biol. 140:313.
33. Ling, G.N. In: Search of the Physical Basis of Life. Plenum Press, New York
and London, 1984.
Full text [PDF]
AuthorÆs remarks made after publication of this article
1. The main phenomenon of this work is illustrated by Fig.2. It is needed to
demonstrate the protein-protein interaction on Fig.2 by some direct method. If
so, it will open a way for further experiments.
2. In our conditions, diffusion of tropomyosin-troponin complex began after 6
hourÆs period after creation of the two-tube systems and came to an end after 12
hours. So, the diffusion takes up about 6 hours.
3. The value of tropomyosin/actin ratio in dAM has important role for
nAM-dAM interaction.
4. In some cases (in 20% of all experiments) a freshly extracted natural
actomyosin has no Ca-sensitivity. All experiments described above were carried
out with natural actomyosin Ca-sensitivity of about 3 a.u.